Get in touch today to see how we can help. That’s why, at ReAgent, we have a skilled and dedicated team who you can speak with about any product enquiry you may have. It is important to understand exactly what you’re purchasing. For example, a 1000ml solution that contains 450ml methanol has a methanol concentration of 45% v/v (450 / 1000 x 100).Īgain, the method for calculating % w/w uses the same steps instead it is weight divided by weight. In the test, a cup is filled with a given quantity of grease. The degree of oil separation will depend upon the thickener, base oil and manufacturing method. 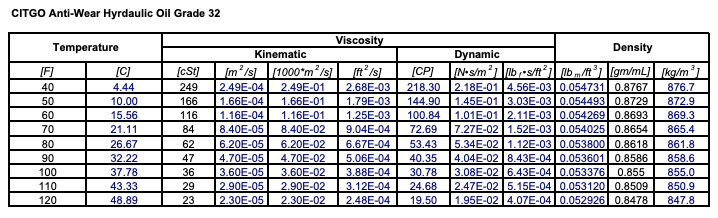
For lubricants, a proper viscosity must helps ensure an adequate separation between surfaces without causing too much friction. When working out the % v/v of a solution, the same method is used except it is the volume of the solute (ml) that is divided by the volume of the solution (ml). A measure of a fluid’s resistance to flow. This tells us that there is a nitric acid solution of 65% w/v. Viscosity is a rating system for oil that rates the oil’s thickness at a given temperature. Therefore, to figure out the % w/v of a 100ml solution that is made up of 65g nitric acid, we would divide 65g by 100ml and then multiply the answer by 100. (Mass of Solute (g) / Volume of Solution (ml)) x 100 For example, to find the % w/v of a solution the calculation is: Calculating PercentagesĮach percentage type can be calculated by making small changes to the same method. If 1g of potassium iodide is used to make up a total volume of 100ml, then a 1% w/v solution of potassium iodide has been made. This is used when a solid chemical is dissolved in a liquid. It can alternatively be abbreviated to m/v for mass per volume. 
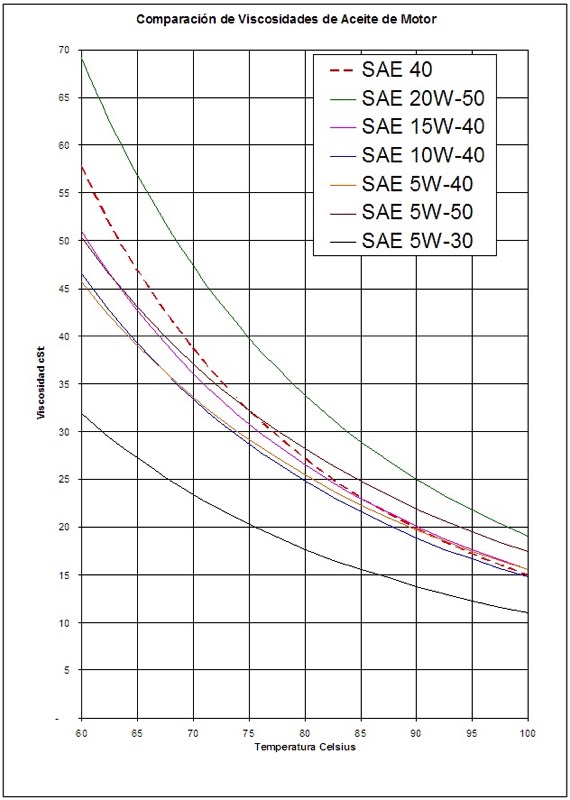
Mass concentration of solution is expressed as % w/v for weight per volume. So, if the total mass of a 100g solution is made up of 30g hydrochloric acid in 70g of water, then we would express this as hydrochloric acid 30% w/w. In this case, the volume of each chemical is disregarded and only the weight is used. Like before, this stands for weight per weight. Weight concentration of a solution is expressed as % w/w. Therefore, we would express the concentration of this solution as sulphuric acid 50% v/v. The Society of Automotive Engineers (SAE) viscosity grading system for engine oils consists of W grades that define low temperature viscosities and. The lower the VI, the more the viscosity is affected by changes in temperature. It is mostly used to characterize the viscosity-temperature behavior of lubricating oils. This is used when both chemicals in a solution are liquid.įor example, when 50ml of sulphuric acid is diluted with 50ml of water, there will be 50ml of sulphuric acid in a total volume of 100ml. The viscosity index ( VI) is an arbitrary, unit-less measure of a fluids change in viscosity relative to temperature change. Volume concentration of a solution is expressed as % v/v, which stands for volume per volume. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
